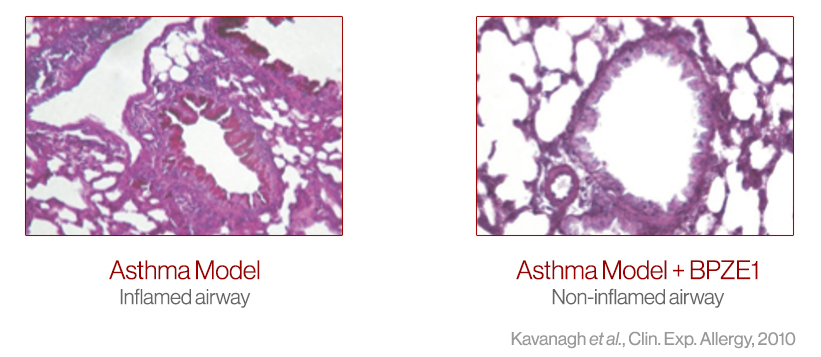
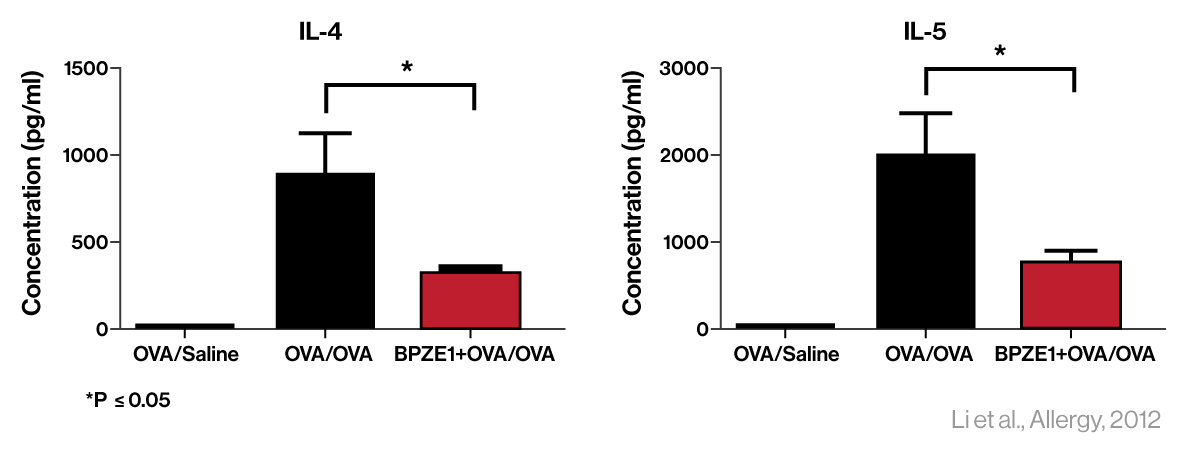
In preclinical studies, prior nasal vaccination with BPZE1 protected against allergic airway inflammation and contact dermatitis in models of human disease (Link) and BPZE1 prevented allergic pulmonary pathology in a mouse model of human asthma (Link). Derived from the B-Tech™ platform, new clinical candidates such as IB-002A are in development to optimize therapeutic allergy effects.

ILiAD is developing protocols for additional B-Tech™ candidate studies to prevent and treat Alzheimer’s-like experimental neurodegenerative disease in mice. These studies will test the effect of an advanced B-Tech™ candidate on markers of cognitive decline, CNS Aβ plaques, and evidence of glial activation.
Due to constant antigenic drift and shift of viral surface proteins, influenza vaccines must be reformulated each year to match the circulating viral subtypes. The B-Tech™ platform has already yielded BPZE1 which has shown significant potential as a universal influenza vaccine due to its ability to reduce pathologic host cytokine responses to infection and, thereby, disease pathology.
BPZE1 has demonstrated effective and sustained protection against lethal challenge with mouse-adapted H3N2 or H1N1 (A/PR/8/34) influenza A viruses. Influenza induces a cytokine storm in serum and lungs caused by uncontrolled activation of the host innate immune system. BPZE1’s mechanism of action is not targeted to reduce viral load; instead BPZE1 dampens the influenza-induced cytokine storm (Link).